Dichloromethane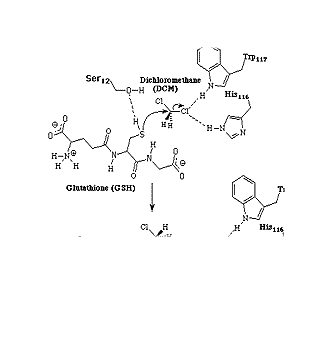
Overview
1.1
What is Dichloromethane?
1.2
Facts and figures
2.1
The Hoechst Process
3.1 Pathway
4.1
Humans
4.2
Animals
1.
Introduction
1.1
What is Dichloromethane
Dichloromethane
(DCM) is a colorless non-flammable liquid. It does not occur naturally and is
produced in high amounts by chemical companies. DCM was introduced as a solvent
over 60 years ago to replace more flammable alternatives. Due to its high
toxicity, recommendations have been issued by the US Environmental Protection
Agency (EPA) to reduce its production. Luckily most producers of this chemical
have complied and the production of this chemical has been continuously reduced
in the past years.
Though
it is biodegradable in soil, it is not readily absorbed in soil, and either
volatilizes into the air or sinks through the soil, into the ground water
table. Exposure to this substance has been shown to result in ill health. As a
result it is a dangerous substance to be dealt with and should be slowly moved
out of the industry.
DCM
is chiefly used an active ingredient in paint
removers (30%), both for consumers and the industry, where it can be
present in concentrations of up to 80% [US Environmental Protection Agency
(EPA)]. Other common uses today are: adhesives
(16%), aerosol and coating (11%),
foam manufacture (10%), chemical processing (9%) and metal cleaning (8%) [Halogenated
Solvent Alliance, Inc.]. It is
also used in chemical processing, where, among other uses, it is used to
extract unwanted substances from foodstuff. It is very efficient, organic,
liquid and inflammable. These qualities and applications have made this
substance a favourite among chemical companies. It can be produced in large
quantities, and once acquired it can be applied to various needs.
1.2 Facts and Figures
[ The
following information is from the EPA ]
CAS No. 75-09-2
Common
Synonyms MC, dichloromethane,
DCM, methylene bichloride,
methylene dichloride
Molecular
Formula CH2Cl2
Chemical Structure
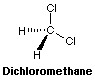
Molecular
Weight 84.94g
Melting
Point -958C
at 101.3 Pa
Boiling
Point 39.758C at 101.3 Pa
Water
Solubility 1.32 x 104 mg/L at 208C
Density 1.3255 g/mL
Vapor Density (air = 1) 2.93
Flash
Point Nonflammable
Odor Threshold 214 ppm (in
air)
Conversion
Factors 1 ppm = 3.48
mg/m3;
1 mg/m3 = 0.288 ppm
2.
Industrial Processes
DCM does not occur naturally and is
produced in large amounts by two main chemical companies in
DCM and tri-chloromethane (TCM) are simultaneously
produced at the same facilities, since the production of DCM produces TCM in
reasonable large amounts. When producing DCM it is important to maintain a high
percent of excess methane in proportion to the chlorine to achieve optimal
yield and to avoid unwanted temperature fluctuations. The high excess of
methane (CH4) also minimizes the concentration of un-reacted
chlorine in the finished process. In addition a very high level of purity with
respect to CH4 has to be observed. Much CH4 is acquired
from secondary processes in other chemical facilities as well distillation of
natural gas using the Linde process.
2.1 The Hoechst Process for DCM
The Hoechst’s process, introduced in 1923,
is still the most common method used today with the mere addition of new
technology but without any modifications of the actual process. CH4
gas and monochlormethane (MCM,
recycled from the previous reaction) is brought into contact with chlorine (Cl2)
gas and both gases are pushed into a reactor in which constant gas circulation
is sustained. The reactor temperature is maintained at 350 – 450 8C (by proper choice
of Cl2 – CH4 /MCM ratio) and the reaction is conducted
adiabatically. The fully reacted mixture
and the resulting hydrogen chloride is then cooled and washed out with dilute
hydrochloric acid in the form 31% hydrochloric acid. Finally the last traces of
HCl(aq) are washed away by the use sodium hydroxide and the
products are largely compressed, dried, cooled, and allowed to condense.
Methane and gaseous MCM are recycled. The liquid is distilled by high pressure
into its principal components – MCM, DCM, TCM and Tetrachloromethane
(TetraCM). This process delivers generally about 70%
DCM by weight. Most of the remainder is TCM with some MCM and trace amounts of TetraCM.
3.
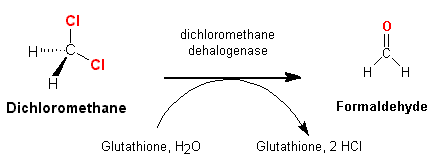
Biodegradation Pathway
As mentioned above, DCM is very volatile
and hence does not stay in surface waters very long. Degradation therefore
occurs in the atmosphere. Aerobic and anaerobic methylotrophic bacteria (methylotrophic
microorganisms are bacteria and fungi which are
growing on carbon compounds which are more reduced than CO2 (i.e.
have less oxygen), [Heinrich Klein,
4. Envioronmental effects <return to top>
DCM enters surface water streams from
industrial effluents and can reach groundwater through underground injection
and through soil. Dichloromethane occurs in soil due to landfills in which
dichloromethane containers have been deposited. It occurs in the air through
evaporation from surface waters, surface soils and from evaporation during use.
Air is inhaled by humans and animals, and surface water is directly used by
animals
Environmental studies have shown that
dichloromethane is highly toxic to humans and animals. In air it has a half
life of approximately 130days [EPA], which is a long time when considering the
quickness with which DCM works. When inhaled it has harmful effects on the
internal systems of animals and humans. It would do harm to the ozone layer,
but it usually undergoes reaction with hydroxyl radicals to form a substance
less harmful to the ozone layer and then continuous its breakdown into other
chemicals. In soil it could biodegrades
slowly, but this sort of biodegradation in soil or subsurface soil only occurs
in landfill sides where there are a substantial amount of microbial populations
(i.e. methylotrphic). Generally its high volatility
forces it to enter the atmosphere. Otherwise it sinks through the soil all the
way to the groundwater. It is as volatile in water as it is in soil, but since
the groundwater table does not have a layer of air on top, it remains in ground
water. In water it hydrolyzes slowly, with a half-life of about 18 months [EPA].
4.1 Effects on humans
DCM is also very harmful before it is
treated as a waste material. As mentioned above it volatilizes readily into the
atmosphere. Hence, workers who use dichloromethane will inevitably inhale air
contaminated with DCM. Since it usually occurs in high concentrations, and has
a high vapour pressure, it poses imminent health risks to all users.
.
DCM is readily absorbed by the lungs and
gastrointestinal tract. Some skin absorption is also existent. DCM, once
absorbed, travels to the liver, kidney, lungs, brains and muscles within one
hour after inhalation according to animal studies reported by the Agency for
Toxic Substances and Disease Registry [ATSDR]. The ATSDR further reports that
within 48hrs dichloromethane expands its spreading to the testes and the epididymal fat.
In all known cases, exposure to DCM has
been shown to affect the nervous system and internal organs in a negative way. It
has been reported that direct contact has led to burns, and over exposure has
led to worker death [EPA]. The Registry of Toxic effect of Chemical Substances [RTECS]
reports that the lowest lethal dose for direct exposure is 357mg/kg, which
translates roughly to 720ppm.
DCM metabolizes to carbon monoxide in
humans, which in turn results in the formation of carboxylhemoglobin
(COHb). COHb deprives the body of oxygen. Dichloromethane, while permanently
damaging the human body by oxygen deprivation, leaves the subject initially in
a state of euphoria, like a drug. Once the concentration of COHb increases, the
subject experiences “psychomotor effects” (e.g. time – interval discrimination)
and cardiovascular changes.
4.2 Effects on animals
Like most chemicals this substance was
tested on animals to verify human reactions. Wild animals are not as exposed to
this chemical as much as humans, as there are not many uses outside the urban
setting. Nonetheless, groundwater and river water can lead to harmful
digestion. Luckily most animals have a keener sense of smell and avoid liquid
containing DCM. Animal studies in rats have resulted in liver damage and kidney
damage after acute exposure (5-6hrs). After chronic exposure the same organs
become the target (2yrs) [RTECS]. It also metabolizes to CO, resulting again COHb.
All studies have shown that dichloromethane
is a dangerous substance to the environment not only as a waste product, but also
during usage. Every worker who handles DCM endangers his health permanently. It
is therefore necessary that strict laws be set in place. DCM should be moved
out of the industry and replaced by less hazardous materials even at the loss
of efficiency and profit. The extreme toxicity of DCM does not allow a truly
save maximum contamination limit. Any maximum contamination limit can be broken
by careless storage or accidents. DCM works fast. Before anyone knows what
exactly had been spilled, workers can be exposed to lethal amounts of DCM.
Deaths and illnesses are hardly worth profit and efficiency, especially in a
legal system that allows workers’ families to sue anybody that can be held
responsible. DCM should therefore be discontinued for both, hidden economical
reason and apparent environmental reasons.
![]()
References:
Ullman’s Encyclopedia of Industrial Chemicals
Chemcial Properties Handbook, McGraw Hill Handbooks
Dierectory of Solvents, Chapman & Hall; Blakcie A&P
U.S. Environmental
Protection Agency , webpage
Agency for Toxic
Substances and Disease Registry , webpage
Registry of Toxic
Effects of Chemical Substances , webpage
Biocatalysis/Biodegradation Database , University of
Michigan, webpage